Then we will go through a brief overview of intermolecular forces, and finally we’ll explore how intermolecular forces govern the way that liquids behave. In this module we’ll first discuss liquids in the context of the other two main states of matter, solids and gases. These varied behaviors arise primarily from the different types of intermolecular forces that are present in liquids. 
You may also notice a similar phenomenon in some salad dressings that separate into an oil layer that rests atop a layer of vinegar, which is primarily water. Consider oil spills, where the oil floats in a sticky, iridescent layer on top of the water. Others, though, don’t seem to mix at all. When it comes to interactions between different liquids, some mix well: Think of a Shirley Temple, made of ginger ale and grenadine. 
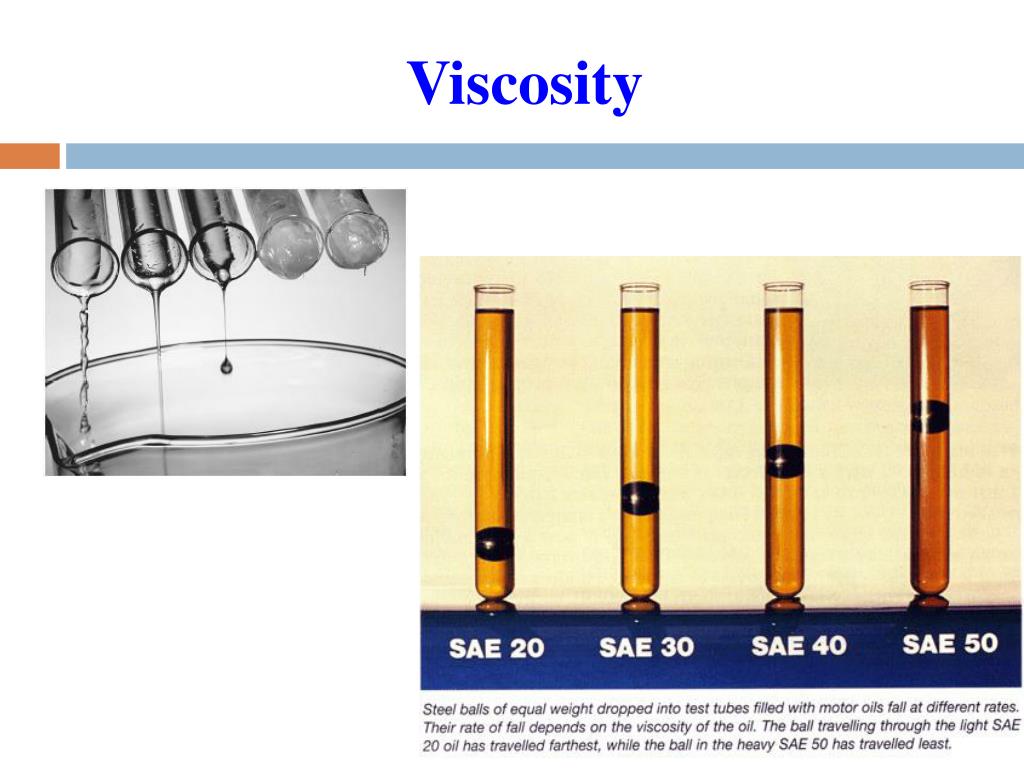
Where do these different behaviors come from? Some liquids flow relatively easily, like water or oil, while others, like honey or molasses, flow quite slowly. Beyond this feature, though, the behaviors of different liquids span a broad range. One of the key defining properties of liquids is their ability to flow. These are just three examples of a highly diverse state of matter: liquids. The viscosity of a liquid decreases with increase in temperature.Intermolecular occurring between different molecules intramolecular occurring within a molecule polar having an electrical charge viscous thick not flowing easily With increase in temperature, the kinetic energy of the molecules of the liquid increases which can overcome the intermolecular forces. Hydrogen bonding and van der waals forces are strong enough to result into high viscosity of the liquid. Water has higher viscosity than methyl alcohol because intermolecular forces in water are greater than those in methyl alcohol. Greater are the intermolecular forces, higher is the viscosity of the liquid. The units of viscosity are dynes sec cm -2.This quantity is called 1 Poise.ġ poise = 1 g cm -1 s-1 = 0.1 kg m -1 s -1Ģ) Effect of nature of the liquid on viscosity This force of friction which one part of the liquid offers to another part of the liquid is called viscosity.Ĭoefficient of viscosity may be defined as the force of friction required to maintain a velocity difference of 1 cm/sec between two parallel layers, 1 cm apart and each having an area of 1 sq cm. In other words, every layer offers some resistance or friction to the layer immediately below it. This type of flow in which there is a regular gradation of velocity in going from one layer to next is called laminarĪs we move from centre towards the walls, the velocity of the layers keep on decreasing. As we move from the wall towards the centre of the tube, the velocity of the cylindrical layers keeps on increasing till it is maximum at the centre. The layers which is in contact with the walls of the tube is almost stationary. Imagine the liquid to be made up of a large number of thin cylindrical coaxial layers. All parts of the liquids do not move through the tube with same velocity. The liquids which flow rapidly have low internal resistance which is due to weak intermolecular forces and hence are said to be less viscous or said to have low viscosity.Ĭonsider a liquid flowing through a narrow tube. The liquids which flow slowly, have high internal resistance which is due to strong intermolecular forces and therefore, are said to be more viscous or are said to have high viscosity. This internal resistance to flow possessed by a liquid is called its viscosity Some liquids like water, ether flow rapidly why some other liquids like glycerine, castor oil flow quite slowly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
